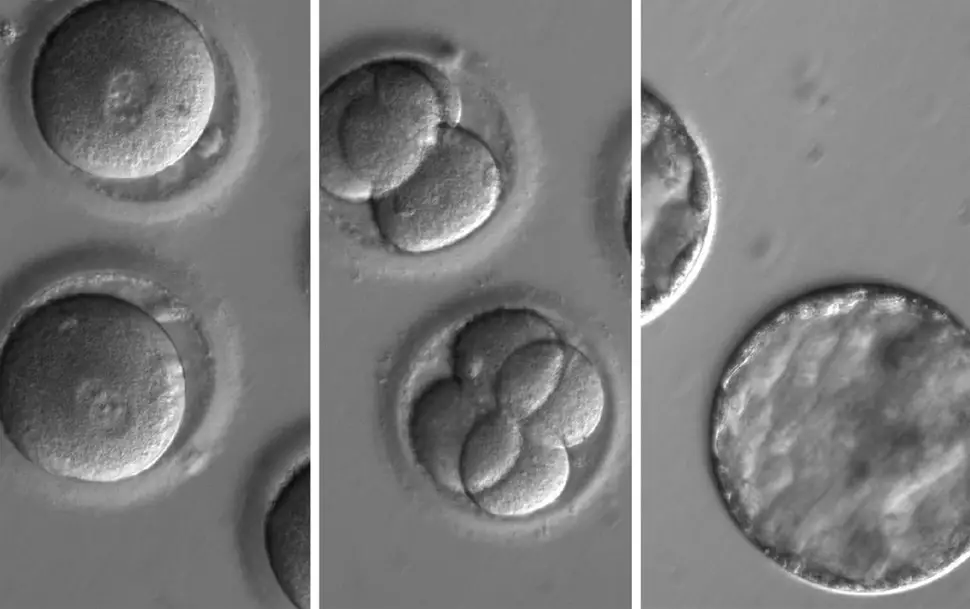
2nd August 2017 CRISPR removes disease from human embryos for the first time For the first time, scientists have used the CRISPR system in human embryos to delete faulty DNA responsible for a hereditary heart condition.
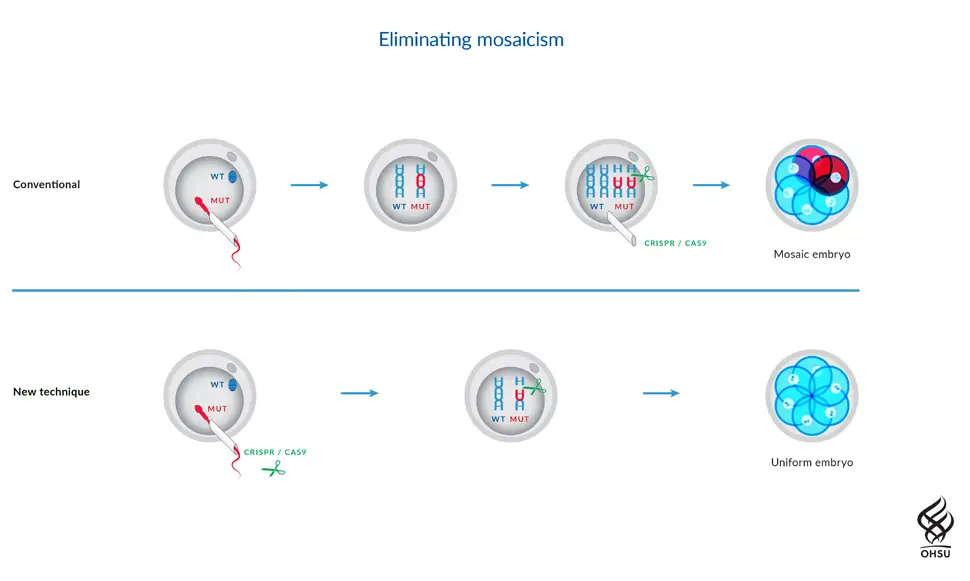
Scientists have, for the first time, corrected a disease-causing mutation in early stage human embryos with gene editing. The technique, which uses the CRISPR-Cas9 system, corrected the mutation for a heart condition at the earliest stage of embryonic development so that the defect would not be passed on to future generations. The work, described today in the journal Nature, is a collaboration between the Salk Institute, Oregon Health and Science University (OHSU) and Korea's Institute for Basic Science. It could pave the way for improved in vitro fertilization (IVF) outcomes, as well as eventual cures for some of the thousands of diseases caused by mutations in single genes. "Thanks to advances in stem cell technologies and gene editing, we are finally starting to address disease-causing mutations that impact potentially millions of people," says Juan Carlos Izpisua Belmonte, a professor in Salk's Gene Expression Laboratory and a corresponding author of the paper. "Gene editing is still in its infancy, so even though this preliminary effort was found to be safe and effective, it is crucial that we continue to proceed with the utmost caution, paying the highest attention to ethical considerations." Hypertrophic cardiomyopathy (HCM) is the most common cause of sudden death in otherwise healthy young athletes, and affects approximately 1 in 500 people overall. It is caused by a dominant mutation in the MYBPC3 gene, but often goes undetected until it is too late. Since people with a mutant copy of the MYBPC3 gene have a 50 percent chance of passing it on to their own children, being able to correct the mutation in embryos would prevent the disease not only in affected children, but also in their descendants. The researchers generated induced pluripotent stem cells from a skin biopsy donated by a male with HCM and developed a gene-editing strategy based on CRISPR-Cas9 that would specifically target the mutated copy of the MYBPC3 gene for repair. The targeted mutated MYBPC3 gene was cut by the Cas9 enzyme, allowing the donor's cells' own DNA-repair mechanisms to fix the mutation during the next round of cell division by using either a synthetic DNA sequence or the non-mutated copy of MYBPC3 gene as a template. Using IVF techniques, the researchers injected the best-performing gene-editing components into healthy donor eggs newly fertilised with the donor's sperm. Then they analysed all the cells in the early embryos at single-cell resolution to see how effectively the mutation was repaired. The scientists were surprised by just how safe and efficient the method was. Not only did a high percentage of embryonic cells get repaired, but also gene correction did not induce any detectable off-target mutations and genome instability – major concerns for gene editing. In addition, the researchers developed a robust strategy to ensure the repair occurred consistently in all the cells of the embryo (spotty repairs can lead to some cells continuing to carry mutations – see illustration below).
Professor Belmonte and his colleagues emphasise that, although promising, these are very preliminary results and more research will need to be done to ensure no unintended effects occur. "Our results demonstrate the great potential of embryonic gene editing, but we must continue to realistically assess the risks as well as the benefits," adds Belmonte. Future work will continue to assess the safety and effectiveness of the procedure and efficacy of the technique with other mutations. However, this latest study has already been condemned by Dr David King, from the campaign group Human Genetics Alert, which described the research as "irresponsible" and a "race for first genetically modified baby". "Perhaps the biggest question, and probably the one that will be debated the most, is whether we should be physically altering genes of an IVF embryo at all," said Darren Griffin, a professor of genetics at the University of Kent. "This is not a straightforward question. Equally, the debate on how morally acceptable it is not to act when we have the technology to prevent these life-threatening diseases must also come into play."
---
Comments »
|